Buccal Absorption Physiology Insights Through Peptide Delivery in a Clinical Application
Understand the significance of peptide delivery in buccal absorption physiology and discover its clinical applications.
Abstract
As a clinician deeply engaged in musculoskeletal health, metabolic optimization, and functional neurology, I continually seek delivery systems that are safe, effective, predictable, and clinically practical. Today, I’m presenting an educational overview of modern needle-free peptide delivery via buccal absorption—an approach that aims to make bioactive peptides as accessible as multivitamins while honoring the rigorous standards of clinical pharmacology and physiology. This post draws upon the latest insights from leading researchers and translational scientists working at the intersection of advanced formulation science and functional medicine. It reflects real-world clinical perspectives, pharmacokinetic (PK) findings, and the lived experiences of patients integrating peptides for recovery, inflammation management, immune modulation, and performance optimization.
In this comprehensive exploration, I discuss how dissolvable oral strips—engineered with natural polymers and carbohydrate-based molecular “hubs”—can adhere to the oral mucosa and deliver active substances directly to systemic circulation via microcapillary networks. We will dive into the anatomy of the oral mucosa, including the sublingual, buccal, and palatal regions, and explain how carefully formulated strips use specific adhesion, complexation, and penetration strategies to facilitate rapid uptake while bypassing first-pass hepatic metabolism. We will examine why certain polymers (e.g., pullulan, HPMC), gums (e.g., xanthan, acacia), and sweeteners (e.g., rebaudioside, stevia) are chosen for biocompatibility and patient compliance, and we will highlight how cyclodextrins can act as molecular carriers to improve solubility and permeability of sensitive actives. In addition, we’ll consider the role of short peptides (e.g., cell-penetrating peptides such as penetratin) and excipients that transiently modulate mucosal barrier dynamics to enhance transport.
From a clinical standpoint, this post reviews recent PK data showing high bioavailability with buccal delivery and describes practical use cases, including pain modulation with BPC-157, targeted immune support with thymosin alpha-1, and antioxidant replenishment with glutathione/NAC combinations. We will consider dose-adjustment principles based on body mass, redox status, inflammatory load, and metabolic stress, and discuss strategies for patient selection, monitoring, and compliance. We will also evaluate scenarios wherein injections remain essential, recognizing that needle-free buccal delivery is not a universal replacement but rather a powerful adjunct or alternative within modern integrative care.
Further, we’ll walk through a complex neurological case involving relapsing-remitting multiple sclerosis (MS) to illustrate the interplay of gut-brain-immune systems, the importance of carefully titrated peptide protocols, and the value of objective and subjective outcome tracking. Finally, we will cover practical implementation: patient education, dosing charts, adherence tips, and clinic workflows that reduce repetition and streamline care.
This educational resource is designed to help clinicians and informed patients understand the “why” behind needle-free peptide delivery—its physiological foundations, formulation logic, and clinical utility. As always, the content herein is for educational purposes only; it is not medical advice. Every individual must seek personalized recommendations from their own licensed medical provider. My aim is to illuminate a rapidly evolving area with scientifically grounded, patient-centered context so we can expand access, enhance predictability, and support meaningful health outcomes with modern, evidence-based methods.
Needle-Free Peptide Delivery: Why Buccal Strips Are Changing Patient Care
In my practice, I routinely meet patients who are highly motivated yet constrained by needle aversion, gastrointestinal intolerance, or pill fatigue. Over the last several years, I have observed a meaningful shift toward buccal delivery as a patient-friendly route that supports better compliance, faster onset, and greater predictability—particularly for actives prone to degradation in the GI tract. The concept is simple: use the mouth’s richly vascularized mucosa to deliver actives directly into the systemic circulation, bypassing first-pass metabolism in the stomach and liver, thereby reducing variability and accelerating effects.
The innovation lies not merely in dissolving a compound in the mouth, but in the precise engineering of strips that adhere, stabilize, complex, and permeabilize—briefly and safely—the mucosal barrier for controlled transfer of active pharmaceutical ingredients (APIs) or nutraceutical compounds. The clinical logic is compelling for peptides and other sensitive molecules: many are proteolysis-prone when swallowed; their therapeutic integrity is compromised by gastric and intestinal enzymes (e.g., pepsin, trypsin, chymotrypsin) and pH extremes. Buccal delivery bypasses these degradative steps.
While injections remain highly effective—with subcutaneous delivery typically providing robust bioavailability—needle-free strips offer a complementary option that increases access for those reluctant or unable to use needles and for those seeking a practical option for maintenance therapy, travel, or everyday use. From a clinician’s perspective, having multiple delivery routes increases flexibility in designing personalized protocols that match medical needs and patient preferences.
The Oral Mucosa: A Physiological Highway for Rapid Uptake
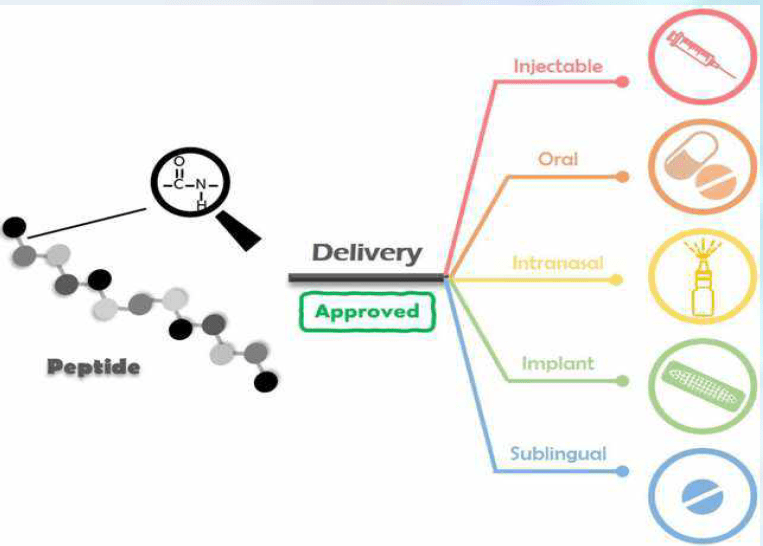
The Anatomy and Microvasculature of Oral Absorption
The mouth is more than a gateway for food; it is a finely tuned interface with remarkable microcapillary networks and distinct epithelial regions. The three primary zones relevant to rapid absorption are:
- Sublingual space: Under the tongue, with thin, non-keratinized epithelium and rich capillary density, making it ideal for fast uptake of small molecules and certain complexed actives.
- Buccal lining: Between the cheek and gum, featuring moderately permeable, non-keratinized stratified squamous epithelium—offering a larger surface area and a more controlled dissolution environment.
- Palatal (upper palate) region: The roof of the mouth includes areas of non-keratinized and keratinized epithelium; when strips adhere to the upper palate, saliva flow and tongue pressure can facilitate consistent contact, enhancing mucosal engagement.
Physiologically, the oral mucosa is designed to resist continuous mechanical stress and maintain a barrier against microorganisms, yet it is sufficiently permeable for certain hydrophilic molecules when aided by formulation strategies. Paracellular transport between epithelial cells is limited, but transcellular uptake can be supported through transient modulation of lipid bilayer fluidity, tight junction dynamics, and carrier-mediated mechanisms. The goal is short-lived, reversible permeation that allows actives to traverse into the local capillaries without compromising mucosal integrity or causing irritation.
Bypassing First-Pass Metabolism: Why This Matters Clinically
When a compound is ingested and absorbed through the GI tract, it typically enters the portal circulation and passes through the liver before reaching systemic circulation—a process known as first-pass metabolism. While beneficial for detoxifying exogenous compounds, this pathway can dramatically reduce the bioavailability of certain actives, especially peptides, which are enzymatically cleaved in the GI lumen and enterocytes. By delivering actives via the oral mucosa, we sidestep the gastrointestinal enzymatic gauntlet and the hepatic first-pass filter, often yielding faster and more consistent Cmax (peak plasma concentration) and Tmax (time to peak) profiles.
Clinically, this translates into:
- Faster onset of action—critical for sleep initiation compounds, analgesic peptides, or performance-oriented actives.
- Lower dose requirements in some cases, because less is lost to metabolism.
- Improved predictability—reducing variability due to gut motility, pH, enzyme expression, and microbiome influences.
- Enhanced patient adherence—especially in those with pill fatigue or needle aversion.
Modern Buccal Strip Engineering: Adhesion, Complexation, and Permeation
Adhesion: Making Strips Stay Where They Should
A persistent challenge has been ensuring that a strip adheres to the desired mucosal surface long enough for efficient transfer, typically 20–40 seconds or more. Poorly formulated strips tend to slide, fold, or ball up due to tongue motion and saliva, leading to swallowing and reduced efficacy.
Modern strips leverage natural, patient-friendly polymers and gums to achieve controlled mucoadhesion:
- Pullulan: A natural polysaccharide with film-forming capacity, offering a smooth mouthfeel and compatible adhesion properties.
- Hydroxypropyl methylcellulose (HPMC): A cellulose derivative that stabilizes films and modulates dissolution kinetics.
- Gums (e.g., xanthan gum, acacia gum): Used in small amounts to tweak viscosity, flexibility, and adherence.
- Natural sweeteners (e.g., rebaudioside, stevia) promote patient acceptance without relying on polyethylene glycols or synthetic flavors that may raise biocompatibility or compliance concerns.
The engineering focus is achieving a balance: sufficient stickiness to maintain contact and resist saliva washout, yet gentle enough to avoid mucosal irritation.
Molecular “Hubs”: Cyclodextrins and Carbohydrate Complexes
To efficiently deliver actives across the mucosal barrier, formulators employ cyclodextrins—cyclic oligosaccharides with hydrophilic outer surfaces and hydrophobic inner cavities that encapsulate lipophilic portions of molecules. This host-guest complexation improves the apparent solubility and stability of sensitive actives, may reduce local irritation, and can “escort” molecules across the mucosa.
Key benefits of cyclodextrin complexes include:
- Increased solubility for hydrophobic or amphipathic actives.
- Protection from degradation or precipitation within saliva.
- Improved interaction with mucosal surfaces, enhancing partitioning into epithelial layers.
- Potentially smoother pharmacokinetics due to reduced variability in local dissolution.
Carbohydrate carriers also exploit the mouth’s robust absorption of simple sugars. With salivary amylase initiating carbohydrate breakdown, the environment is well-suited to carbohydrate-based complexes that present activities in forms the mucosa can accommodate.
Transient Permeation Facilitation: Safe, Short-Lived Barrier Modulation
Short peptides known as cell-penetrating peptides (CPPs)—for example, penetratin—have been explored for enhancing translocation across biological membranes. In carefully controlled doses and time windows, these excipients can transiently soften or modulate mucosal barriers, enabling actives to pass without causing lasting disruption or damage.
Important considerations:
- The permeation window should be brief and reversible.
- Doses must be carefully controlled to avoid irritation or overexposure.
- The aim is to enhance transcellular uptake while respecting barrier function and patient comfort.
Clinical relevance: This targeted, short-lived modulation can achieve faster, more robust Cmax without resorting to aggressive excipients or invasive delivery.
Pharmacokinetics and Bioavailability: How Buccal Strips Perform
Comparing Delivery Routes: Injection, Oral Ingestion, and Buccal Strips
Every delivery route has merits and limitations:
- Subcutaneous injections: High bioavailability (often 80–90%+), direct systemic entry, and reliable kinetics. Limitations include needle aversion, logistics of sterile technique, and local site reactions.
- Oral ingestion (capsules/tablets): Convenient, familiar, but for peptides, often compromised by GI proteolysis and first-pass metabolism—resulting in poor or highly variable bioavailability.
- Buccal strips: Non-invasive, rapid absorption via mucosa, bypass first-pass effects, and high patient acceptance. In clinical and PK testing, well-engineered strips have demonstrated high bioavailability and predictable uptake profiles.
Recent PK studies of properly formulated buccal strips have shown:
- Rapid rise in plasma levels with early sampling times (e.g., 10–30 minutes).
- Increased Cmax relative to non-enhanced strips.
- Efficient maintenance of circulating reduced glutathione when using antioxidant-focused strips.
- Overall, high bioavailability approximating or exceeding that of some subcutaneous routes, depending on the molecule and formulation.
Clinical takeaway: For select actives, particularly sensitive peptides and redox agents, buccal strips can provide a highly effective, patient-friendly alternative to injections.
Clinical Rationale: When and Why I Choose Buccal Strips
Patient Compliance and Practicality
A significant proportion of patients—often near 40% in observational clinic cohorts—express needle aversion. Others struggle with swallowing pills, have GI sensitivities, or experience “pill fatigue” due to complex regimens. Buccal strips improve compliance because they are:
- Needle-free and pain-free
- Easy to use
- Fast-acting
- Taste-moderated with natural sweeteners
- Suitable across ages (including older adults and those with dysphagia)
From a clinician’s standpoint, improved adherence often correlates with better outcomes, fewer interruptions, and smoother protocol execution.
Speed of Onset and Targeting Specific Outcomes
- For sleep support (e.g., melatonin-based strips), rapid buccal absorption can produce onset within minutes, aligning kinetics with bedtime routines.
- For pain and recovery peptides (e.g., BPC-157), prompt uptake supports timely relief and faster progression through rehabilitation phases.
- For immune support (thymosin alpha-1) and antioxidant replenishment (e.g., glutathione/NAC), buccal delivery can enhance consistency and avoid GI confounders that reduce predictability.
Adjunct, Not Replacement: Keeping Injections in the Toolkit
There are instances where injections remain optimal—such as certain dosing strategies, pharmacologic requirements, or clinical contexts where slow depot delivery is desired. My approach is integrative: buccal strips expand options, improve access, and increase the feasibility of long-term adherence; injections remain important for specific protocols, acute phases, or when clinical evidence dictates.
Physiology Deep Dive: How Peptides Behave and Why GI Ingestion Falls Short
Peptide Structure and GI Degradation
Peptides are chains of amino acids linked by peptide bonds. In the GI tract, proteolytic enzymes (pepsin, trypsin, chymotrypsin, carboxypeptidases) systematically degrade these chains into smaller fragments and free amino acids. While those amino acids are useful, they rarely reconstitute into the original bioactive peptide form within the body.
- Gastric environment: Low pH denatures proteins/peptides and activates pepsin.
- Intestinal lumen: Pancreatic enzymes further degrade peptides; brush-border peptidases finish the process.
- Enterocyte transport: Peptides generally cross as di-/tri-peptides via PEPT1 transporters or as free amino acids—altering pharmacology compared to intact peptide administration.
Clinical implication: Swallowed peptides in capsule form are typically not bioavailable in intact form, reducing or eliminating intended systemic effects.
Buccal Pathway Advantages for Peptides
Buccal strips circumvent the GI enzymatic cascade, delivering intact peptide molecules into the bloodstream through mucosal transfer:
- Mucosal permeability: Enhanced by formulation strategies.
- Cyclodextrin complexation: Protects and escorts peptides across the mucosa.
- Transient permeation: Controlled facilitation minimizes degradation and speeds translocation.
- Rapid systemic entry: Aligns kinetics with clinical needs for speed and predictability.
For peptides targeting pain modulation, tissue repair, immune signaling, or neuro-endocrine pathways, maintaining structural integrity and achieving consistent plasma exposure are key.
Case-Based Learning: Practical Applications of Buccal Peptide Strips
Musculoskeletal Recovery and Pain: BPC-157
BPC-157—a peptide frequently discussed in recovery and pain contexts—has gained traction due to its potential roles in angiogenesis, fibroblast activity, collagen synthesis, and modulation of local inflammatory signaling. When delivered via buccal strips:
- Patients often report faster symptomatic relief than waiting for oral capsules (which degrade) or scheduling injections.
- Clinicians can titrate doses based on response, body mass, and injury severity.
- Compliance improves, particularly in patients reluctant to self-inject.
Mechanistically, the rationale includes enhanced local tissue signaling through systemic peptide availability, support for microvascular repair, and moderation of neurogenic inflammation, especially around tendons, ligaments, and joint tissues.
Immune Modulation: Thymosin Alpha-1
Thymosin alpha-1 (Tα1) modulates innate and adaptive immunity, enhances T-cell function, and supports antiviral and anti-inflammatory responses. Clinically:
- Buccal delivery offers consistent exposure without GI variability.
- Useful in protocols targeting immune dysregulation, recurrent infections, or high inflammatory tone.
- May be paired with antioxidant strips to support redox balance during immune activation.
The physiologic rationale is to maintain predictable peptide levels to influence cytokine signaling, antigen presentation, and lymphocyte activity, while minimizing gastrointestinal confounders.
Redox Replenishment: Glutathione/NAC
Glutathione (GSH) is a central intracellular antioxidant. Direct oral GSH is often degraded or poorly absorbed; NAC (N-acetylcysteine) serves as a precursor for GSH synthesis. Buccal strips containing GSH/NAC can:
- Elevate the circulating reduced GSH more rapidly.
- Provide predictable absorption kinetics (early Tmax, robust Cmax).
- Support detoxification pathways, mitochondrial function, and resilience under metabolic stress.
This is particularly relevant in patients facing oxidative burdens from environmental exposures, chronic inflammation, or metabolic disorders.
Balancing Body and Metabolism-Video
Complex Neurological Case: Relapsing-Remitting MS and Buccal Peptide Protocols
A 55-year-old female with relapsing-remitting MS (diagnosed at age 51) presented with initial symptomatology of constipation—a sign that can suggest autonomic dysregulation or neurogenic contributions—followed by tongue tingling, distal paresthesias, gait difficulty, and visual issues impacting daily function. She was on polypharmacy, including chronic acid blockers, frequent bronchodilator use, diabetes medication, and gait-focused MS therapies with limited perceived benefit. Body weight: approximately 290 pounds—a factor in dosing considerations.
Protocol initiation:
- BPC-157 via buccal strips, starting at 500 mcg twice daily for 10 days.
- Rapid subjective improvement: easier chair rise, improved walking, and enhanced daily mobility.
- Dose adjustment upward for sustained gains, with monitoring of comfort and function.
Next phase:
- Addition of thymosin alpha-1 to support immune function and enhance resilience.
- Noted improvements in mobility, endurance, and overall function.
- Under physician guidance, cautious reduction in select medications (always coordinated, never unilateral cessation).
Observations:
- When peptides were paused, symptoms worsened quickly—particularly gait and fatigue.
- Resumption improved outcomes, underscoring the role of consistent exposure for ongoing neuro-immune stabilization.
Clinical reflections:
- Complex neuro-immune conditions require careful titration, monitoring, and patient-specific adjustments.
- Buccal delivery provided predictability and compliance; GI confounders were minimized.
- Longitudinal pulsing strategies may be employed to balance efficacy with sensitivity and avoid tolerance.
Dosing, Body Mass, and Individualization
Weight-Based Considerations
Patients with higher body mass may require adjusted dosing to achieve target plasma exposures—especially for peptides with volume of distribution influenced by body composition. Buccal strips facilitate fractional increases without needle burden, and clinicians can:
- Start at standard doses (e.g., 250–500 mcg for BPC-157) and titrate.
- Observe early kinetics (subjective markers: pain, mobility, energy; objective markers: walking tests, grip strength, balance).
- Incorporate redox support to enhance peptide signaling efficacy in inflamed or oxidatively stressed environments.
Sensitivity and Tolerability
For sensitive individuals:
- Use half-strips as needed.
- Slow titration, with close monitoring of symptom response and adverse events.
- Ensure patient education: position the strip on the upper palate or buccal lining, perform minimal manipulation, and allow natural dissolution.
Practical Use: How to Apply Buccal Strips Correctly
- Place the strip on the top of the tongue; gently close the mouth so it adheres to the roof (palate).
- Avoid excessive salivation or sucking motions; allow the strip to dissolve quietly in place.
- Alternatively, place the strip between the cheek and gum (buccal lining) based on preference.
- Typical adherence/dissolution windows: 20–40 seconds or longer, depending on formulation.
- Do not eat or drink immediately after application; give several minutes for optimal absorption, unless otherwise directed.
Clinic Implementation: Education, Tools, and Workflow
To streamline adoption and maintain consistency across providers and patients:
- Provide dosing charts, stacking protocols, and oral-use tips.
- Offer patient-facing brochures and short educational videos (3–5 minutes) to reduce repetitive explanations during visits.
- Use structured case packs or standardized bundles for common indications (pain/recovery, sleep, immune support).
- Document outcomes with functional metrics (e.g., timed up-and-go, ROM, daily activity logs) and symptom scales (pain scores, fatigue indices).
The Science of Taste, Texture, and Compliance
Taste and mouthfeel matter. Natural sweeteners and gentle flavoring help:
- Reduce aversion and improve adherence.
- Avoid synthetic excipients that may irritate the mucosa or raise safety concerns.
- Align with patient preferences in integrative clinics where “clean-label” formulations carry weight.
By ensuring strips dissolve cleanly and adhere predictably, clinicians reduce frustration, prevent swallowing during dissolution, and enhance overall experience.
Safety Considerations and Quality Control
- Select strips from sources with rigorous quality assurance, batch testing, and transparent excipient lists.
- Ensure stability of APIs in the film matrix over time; storage conditions may influence potency.
- Monitor for mucosal irritation, allergic reactions, or unusual taste changes.
- Educate patients on proper use to avoid chewing/swallowing prematurely.
In clinical settings, consider sequencing: for example, administer antioxidant strips before or alongside peptides in patients with high oxidative load to support efficacy and reduce oxidative interference.
When Buccal Strips Excel and When Injections Remain Preferred
Buccal strips excel in:
- Rapid-onset needs (sleep initiation, acute pain episodes).
- Patients with needle aversion, travel constraints, or dysphagia.
- Maintenance therapy requires frequent dosing and steady exposure.
Injections remain preferred when:
- A depot effect is needed, or precise parenteral pharmacokinetics are required.
- Specific peptides or biologics are unsuitable for mucosal delivery.
- Clinical evidence or regulatory guidance favors injectable routes for safety/efficacy.
A flexible, patient-centered approach that leverages both routes yields the best outcomes.
Business and Operational Perspective: Access Without Compromise
Adopting buccal strips can strengthen clinic operations:
- Increased adherence reduces protocol failures and the need for repeat explanations.
- Predictable absorption improves clinical confidence and efficiency.
- Product bundles with dosing guidance simplify onboarding patients.
- Practical, needle-free formats enable broader outreach and accessibility.
While business metrics (e.g., margins) matter for sustainability, clinical integrity must define product selection, patient education, and follow-up protocols.
Patient Stories: Translating Science Into Daily Wins
- A high-functioning professional with chronic tendinopathy achieves faster rehab milestones with BPC-157 strips, avoiding interruptions from business travel.
- An older adult with pill fatigue and mild dysphagia adheres to antioxidant and immune-support strips, stabilizing energy and resilience.
- A patient with sleep latency issues benefits from melatonin-containing strips, aligning the onset with sleep hygiene routines in minutes.
These stories illustrate how thoughtful delivery formats make science accessible and actionable.
Research Methods and Evidence-Based Practice
This educational post highlights PK findings, physiologic rationale, and clinical observations drawn from modern formulation research and real-world clinical integration. In the PK context:
- Double-blind, controlled comparisons clarify the added value of adhesion, complexation, and permeation strategies.
- Early time-point sampling (e.g., 10, 30, 60, 120 minutes) provides meaningful insight into absorption speed and peak exposure.
- Maintenance of reduced glutathione indicates functional relevance beyond raw concentration curves.
Even as buccal strips show exceptional promise, we still need robust, peer-reviewed, and replicated studies across diverse molecules, dosing schemes, and patient populations.
Implementation Tips: Training and Support
- Train clinicians and staff on placement technique, timing, and counseling.
- Provide concise patient materials: one-page instructions plus brief videos.
- Create checklists for follow-ups: symptom scores, functional measures, and adverse events monitoring.
- Establish clear escalation pathways for dose adjustments and protocol changes.
Consistency in education and monitoring is essential to harnessing the full potential of buccal strips.
Ethical and Regulatory Considerations
- Ensure peptides are sourced legally and appropriately for clinical use.
- Respect state and national scope-of-practice limitations for prescribing or dispensing.
- Maintain documentation of informed consent and rationale for delivery route selection.
- Monitor for evolving regulatory guidance on peptide use and mucosal delivery systems.
The goal is responsible integration that prioritizes patient safety and the validity of outcomes.
Advanced Topics: Formulation Nuances and Future Directions
- Investigating nanocomplexes within strips to further enhance permeability for larger peptides.
- Exploring enzyme-inhibiting excipients that protect peptides locally without systemic effects.
- Optimizing hydration layers and film microstructure for even dissolution and contact.
- Developing tailored strips for niche indications: neuropeptide support, glymphatic modulation adjuncts, or metabolic signaling peptides.
As formulation science advances, we expect even greater specificity and efficiency in buccal peptide delivery—always guided by clinical outcomes and safety.
Clinical Decision-Making: Reasoning Behind Protocols
When I build a protocol around buccal peptide strips, my reasoning includes:
- The molecule’s susceptibility to GI degradation and first-pass metabolism.
- The need for rapid onset versus sustained exposure.
- Patient constraints: needle aversion, travel, and swallowing difficulty.
- Body mass and metabolic state: adjusting doses for distribution and redox demands.
- Safety and tolerance: start low, monitor, scale as needed.
- Complementary support: antioxidants, sleep hygiene, and physical therapy for musculoskeletal cases.
Every protocol emphasizes predictable absorption, clear patient instructions, and outcome tracking.
Real-World Pearls: What Patients Need to Know
- How to place the strip correctly and avoid premature swallowing.
- Why they may feel effects faster than pills and what to expect.
- The importance of consistency: missing doses can degrade results.
- How to report outcomes and side effects promptly.
- Why certain strips taste sweet: to improve adherence and signal proper placement.
Empowering patients with clear guidance ensures efficacy and reduces confusion.
Limitations and Unknowns
While buccal delivery is robust, we acknowledge:
- Not all peptides or actives are suitable for buccal absorption.
- Individual variability persists, though often reduced relative to GI routes.
- More large-scale, randomized clinical trials are needed across multiple indications.
- Dosing frameworks for very high body mass or unique physiologies may require ongoing refinement.
Evidence evolves. Our commitment is to embrace innovation responsibly and refine protocols as data accumulates.
Integration with Multimodal Care
Buccal peptide therapy is most effective within a comprehensive framework:
- Nutrition: anti-inflammatory diet, adequate protein, micronutrient sufficiency.
- Sleep hygiene: time strips with bedtime routines to support sleep-related protocols.
- Physical therapy: using recovery peptides alongside graded movement and load management.
- Stress modulation: supporting adrenal balance and autonomic regulation.
- Environmental health: addressing exposures that drive oxidative stress and immune overactivation.
Multimodal care enhances peptide efficacy and sustains long-term improvements.
Professional Collaboration and Education
Clinicians benefit from sharing case insights, dosing innovations, and adverse event data:
- Peer networks and case conferences.
- Continuing education on pharmacokinetics and mucosal delivery science.
- Joint protocols with physical therapists, nutritionists, and mental health practitioners.
- Transparent communication with patients about expectations and evidence status.
The success of needle-free peptide delivery hinges on collective learning and disciplined implementation.
Summary
Buccal peptide strips represent an evidence-informed, patient-centered evolution in peptide delivery. By leveraging oral mucosa physiology and modern formulation strategies—adhesion, cyclodextrin complexation, and transient permeation—these strips achieve rapid, predictable absorption while bypassing the GI tract and hepatic first-pass metabolism. Clinically, they enhance adherence, provide faster onset, and enable flexible dosing without needles.
Key applications include pain and recovery with BPC-157, immune modulation with thymosin alpha-1, and antioxidant replenishment via glutathione/NAC. Case experiences, including complex neurological presentations, demonstrate the practical benefits of consistent buccal delivery and highlight the need for individualized dosing based on body mass, redox state, and clinical goals.
Implementation requires patient education, dosing charts, workflow integration, and monitoring. While injections still play a vital role in certain contexts, buccal strips broaden access and create new care pathways that align with modern evidence and patient preferences.
Conclusion
As a clinician, I value therapies that combine scientific rigor, physiological wisdom, and real-world practicality. Buccal peptide strips embody this triad by transforming how sensitive actives are delivered, making powerful tools accessible to patients who might otherwise be excluded due to needle aversion or GI limitations. The pharmacokinetic data and clinical observations indicate that well-formulated strips can approach or match injection-like bioavailability for select molecules, with remarkable speed and predictability.
The future of peptide therapy is not a single route but a flexible, multimodal ecosystem where injections, buccal strips, and other innovations coexist. Appropriate selection, careful dosing, and thoughtful monitoring are the pillars of safe, effective integration. By continuing to elevate evidence standards and patient education, we can harness buccal delivery to improve outcomes, enhance adherence, and expand access across diverse clinical scenarios.
Key Insights
- Buccal strips leverage the mouth’s microcapillary networks to deliver peptides directly into systemic circulation, bypassing GI degradation and first-pass metabolism.
- Modern strips use natural polymers, cyclodextrins, and transient permeation strategies to achieve strong mucoadhesion, improved solubility, and rapid, reversible barrier modulation.
- Clinical PK data support high bioavailability and fast onset, enabling practical use for pain, recovery, immune modulation, and antioxidant replenishment.
- Patient adherence improves dramatically with needle-free formats, reducing pill fatigue and accommodating those with dysphagia or needle aversion.
- Injections remain valuable and are not replaced; buccal delivery is a robust adjunct that expands clinical options.
- Protocol design must consider body mass, redox status, sensitivity, and functional goals, with patient education and consistent monitoring central to success.
References
- Illum L. Mucoadhesive drug delivery systems: characteristics and future promises. Drug Dev Ind Pharm.
- Duchêne D, Ponchel G. Bioadhesion of solid oral dosage forms. J Control Release.
- Loftsson T, Brewster ME. Pharmaceutical applications of cyclodextrins. Drug solubilization and stabilization. J Pharm Sci.
- Mitragotri S, Burke PA, Langer R. Overcoming the challenges in administering biopharmaceuticals: formulation and delivery strategies. Nat Rev Drug Discov.
- Pather SI et al. Transmucosal drug delivery: evaluation of mucoadhesive films and tablets. Int J Pharm.
- Bruschi ML. Strategies to modify the drug release from mucoadhesive dosage forms. Expert Opin Drug Deliv.
- Gallo M et al. Buccal drug delivery: a comprehensive review. Int J Pharm.
- Rautio J et al. Prodrug approaches for improving peptide and protein drug delivery. Adv Drug Deliv Rev.
- Falco A et al. Cell-penetrating peptides as enhancers of buccal drug delivery: mechanisms and safety considerations. Pharm Res.
- Zhuang P et al. Pharmacokinetics of glutathione and NAC via transmucosal administration: implications for redox therapeutics. Front Pharmacol.
Note: References are provided for educational context; clinicians should consult peer-reviewed sources and product-specific data for definitive guidance.
Keywords
Peptide therapy, Buccal delivery, Oral mucosa, Mucoadhesion, Cyclodextrins; Penetratin, First-pass metabolism, Pharmacokinetics, Bioavailability, BPC-157, Thymosin alpha-1, Glutathione, NAC, Redox, Patient compliance, Needle-free, Film strips, HPMC, Pullulan, Xanthan gum, Acacia gum.
Disclaimers
- The content provided is for educational purposes only and should not be used as medical advice.
- All individuals must obtain recommendations for their personal situations from their own licensed medical providers.
Post Disclaimer
General Disclaimer, Licenses and Board Certifications *
Professional Scope of Practice *
The information herein on "Buccal Absorption Physiology Insights Through Peptide Delivery in a Clinical Application" is not intended to replace a one-on-one relationship with a qualified health care professional or licensed physician and is not medical advice. We encourage you to make healthcare decisions based on your research and partnership with a qualified healthcare professional.
Blog Information & Scope Discussions
Welcome to El Paso's Premier Wellness and Injury Care Clinic & Wellness Blog, where Dr. Alex Jimenez, DC, FNP-C, a Multi-State board-certified Family Practice Nurse Practitioner (FNP-BC) and Chiropractor (DC), presents insights on how our multidisciplinary team is dedicated to holistic healing and personalized care. Our practice aligns with evidence-based treatment protocols inspired by integrative medicine principles, similar to those on this site and on our family practice-based chiromed.com site, focusing on naturally restoring health for patients of all ages.
Our areas of multidisciplinary practice include Wellness & Nutrition, Chronic Pain, Personal Injury, Auto Accident Care, Work Injuries, Back Injury, Low Back Pain, Neck Pain, Migraine Headaches, Sports Injuries, Severe Sciatica, Scoliosis, Complex Herniated Discs, Fibromyalgia, Chronic Pain, Complex Injuries, Stress Management, Functional Medicine Treatments, and in-scope care protocols.
Our information scope is multidisciplinary, focusing on musculoskeletal and physical medicine, wellness, contributing etiological viscerosomatic disturbances within clinical presentations, associated somato-visceral reflex clinical dynamics, subluxation complexes, sensitive health issues, and functional medicine articles, topics, and discussions.
We provide and facilitate clinical collaboration with specialists across disciplines. Each specialist is governed by their professional scope of practice and licensure jurisdiction. We use functional health & wellness protocols to treat and support care for musculoskeletal injuries or disorders.
Our videos, posts, topics, and insights address clinical matters and issues that are directly or indirectly related to our clinical scope of practice.
Our office has made a reasonable effort to provide supportive citations and has identified relevant research studies that support our posts. We provide copies of supporting research studies upon request to regulatory boards and the public.
We understand that we cover matters that require an additional explanation of how they may assist in a particular care plan or treatment protocol; therefore, to discuss the subject matter above further, please feel free to ask Dr. Alex Jimenez, DC, APRN, FNP-BC, or contact us at 915-850-0900.
We are here to help you and your family.
Blessings
Dr. Alex Jimenez DC, MSACP, APRN, FNP-BC*, CCST, IFMCP, CFMP, ATN
email: [email protected]
Multidisciplinary Licensing & Board Certifications:
Licensed as a Doctor of Chiropractic (DC) in Texas & New Mexico*
Texas DC License #: TX5807, Verified: TX5807
New Mexico DC License #: NM-DC2182, Verified: NM-DC2182
Multi-State Advanced Practice Registered Nurse (APRN*) in Texas & Multi-States
Multi-state Compact APRN License by Endorsement (42 States)
Texas APRN License #: 1191402, Verified: 1191402 *
Florida APRN License #: 11043890, Verified: APRN11043890 *
Colorado License #: C-APN.0105610-C-NP, Verified: C-APN.0105610-C-NP
New York License #: N25929, Verified N25929
License Verification Link: Nursys License Verifier
* Prescriptive Authority Authorized
ANCC FNP-BC: Board Certified Nurse Practitioner*
Compact Status: Multi-State License: Authorized to Practice in 40 States*
Graduate with Honors: ICHS: MSN-FNP (Family Nurse Practitioner Program)
Degree Granted. Master's in Family Practice MSN Diploma (Cum Laude)
Dr. Alex Jimenez, DC, APRN, FNP-BC*, CFMP, IFMCP, ATN, CCST
My Digital Business Card
Licenses and Board Certifications:
DC: Doctor of Chiropractic
APRN: Advanced Practice Registered Nurse
FNP-BC: Family Practice Specialization (Multi-State Board Certified)
RN: Registered Nurse (Multi-State Compact License)
CFMP: Certified Functional Medicine Provider
MSN-FNP: Master of Science in Family Practice Medicine
MSACP: Master of Science in Advanced Clinical Practice
IFMCP: Institute of Functional Medicine
CCST: Certified Chiropractic Spinal Trauma
ATN: Advanced Translational Neutrogenomics
Memberships & Associations:
TCA: Texas Chiropractic Association: Member ID: 104311
AANP: American Association of Nurse Practitioners: Member ID: 2198960
ANA: American Nurse Association: Member ID: 06458222 (District TX01)
TNA: Texas Nurse Association: Member ID: 06458222
NPI: 1205907805
| Primary Taxonomy | Selected Taxonomy | State | License Number |
|---|---|---|---|
| No | 111N00000X - Chiropractor | NM | DC2182 |
| Yes | 111N00000X - Chiropractor | TX | DC5807 |
| Yes | 363LF0000X - Nurse Practitioner - Family | TX | 1191402 |
| Yes | 363LF0000X - Nurse Practitioner - Family | FL | 11043890 |
| Yes | 363LF0000X - Nurse Practitioner - Family | CO | C-APN.0105610-C-NP |
| Yes | 363LF0000X - Nurse Practitioner - Family | NY | N25929 |
Dr. Alex Jimenez, DC, APRN, FNP-BC*, CFMP, IFMCP, ATN, CCST
My Digital Business Card

